[Source: Supplied]
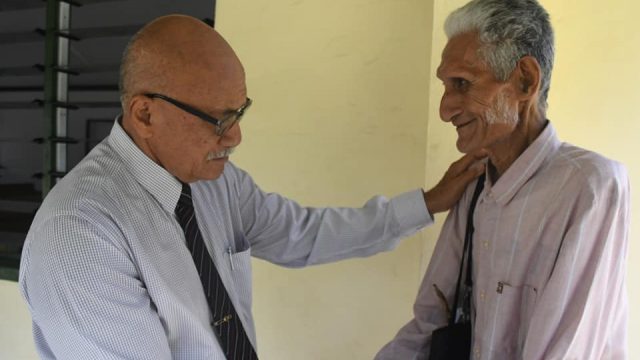
The Ministry of Trade will enter into a three-year memorandum of understanding with Aether Pacific Pharmaceuticals Ltd trading as Medical Kiwi for the development of medicinal cannabis in Fiji.
The MOU will be done to establish a framework for collaboration between Medical Kiwi and the Government.
Medical Kiwi is a registered company in New Zealand.
It is a market leader in New Zealand, in the cultivation and export of medicinal cannabis and cannabidiol to export markets using innovative science and technology.

[Source: Supplied]
Trade Minister, Manoa Kamikamica, explains the reason behind working with Medical Kiwi.
“We need to have investors that actually fits in the business, and have the necessary expertise. So that is one of things we will ensure that happens so that we don’t lose control when we roll out the industry.”
Medical Kiwi intends to build an extraction and manufacturing facility in Fiji.
The facility is aimed to eventually obtain the Good Manufacturing Practices certification for the nutraceutical and pharmaceutical markets.
This comes after the cabinet approved the Policy Framework to establish the medicinal cannabis industry in Fiji after receiving an update on the progress made by the Taskforce.
The Taskforce was appointed in August last year to carry out national and community consultations on the proposal.
The Taskforce was also mandated to oversee the feasibility of this industry and assess the laws required for this emerging market.
It carried out media and private sector engagements, as well as public and community consultations.
When the medical cannabis industry is established by law and policy in Fiji for cultivation and export, Medical Kiwi will still be required to apply for the appropriate license and adhere to the rules and regulations stipulated.
Medical Kiwi endeavours to invest a minimum of $65 million within three to five years, subject to the extent permissible by law.
Cabinet has noted that the Market Development Facility will fund the feasibility study.

 Ritika Pratap
Ritika Pratap