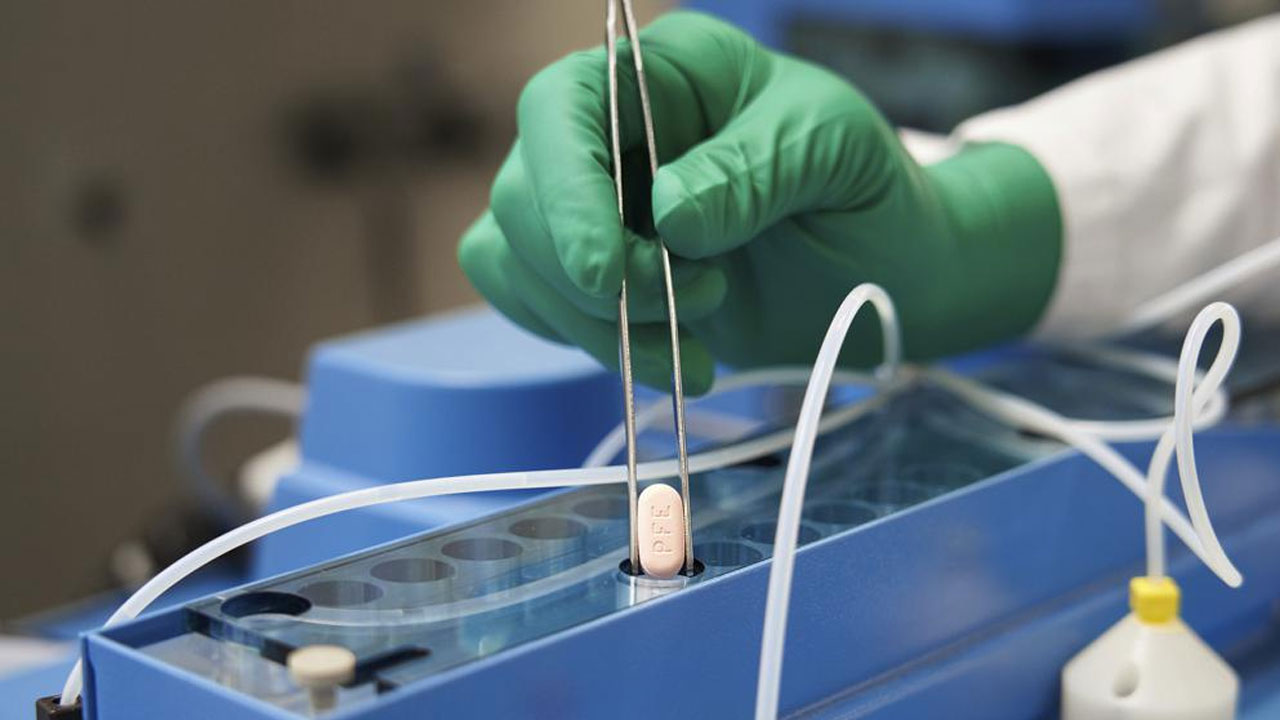
This image provided by Pfizer shows the company's COVID-19 Paxlovid pills. [Source: Pfizer via AP]
Two brand-new COVID-19 pills that were supposed to be an important weapon against the pandemic in the U.S. are in short supply and have played little role in the fight against the omicron wave of infections.
The problem, in part, is that production is still being ramped up and the medicines can take anywhere from five to eight months to manufacture.
While the supply is expected to improve dramatically in the coming months, doctors are clamoring for the pills now, not just because omicron is causing an explosion of cases but because two antibody drugs that were once the go-to treatments don’t work as well against the variant.
“This should be a really joyous time because we now have highly effective antiviral pills,” said Erin McCreary, a pharmacist and administrator at the University of Pittsburgh Medical Center. “Instead, this feels like the hardest and most chaotic stretch of the pandemic.”
The pills — and other COVID-19 drugs, for that matter — are being carefully rationed, reserved for the highest-risk patients.
“January is going to be a terrible month with a million cases a day,” said University of North Carolina virologist Dr. Myron Cohen. “Most people will do perfectly well, but we have to select out the people who won’t and give them the drugs we have available.”
The Food and Drug Administration authorized the two pills from Pfizer and Merck late last month based on studies showing they cut the risk of severe disease and death when given shortly after symptoms appear. They are the first COVID-19 treatments patients can take at home, and thus could reduce the burden on hospitals.
The U.S. didn’t make the kind of mass purchases in advance that it did with vaccines.
Pfizer’s pill, Paxlovid, is considered far superior to Merck’s, but because of the six to eight months it takes to manufacture, the company says it can supply only about 250,000 courses of the treatment by the end of this month.
The U.S. has ordered enough Paxlovid to treat 20 million people, but the first 10 million orders won’t be delivered until June.
White House COVID-19 coordinator Jeff Zients said this week that the government collaborated with Pfizer to help speed up development of the pill by several months, and that officials continue to work with the company to look for ways to boost production.
Pfizer said it is adding capacity: “We expect to use our strong manufacturing capabilities and our extensive supplier network to continue to improve output rapidly.”
Merck’s pill, molnupiravir, was produced in greater advance quantities. It takes six months to manufacture, though the company says that can be compressed to five months if necessary.
But final testing showed the drug was far less effective than Pfizer’s pill and carried significant risks, including the potential for birth defects when taken by pregnant women. As a result, it is considered the last-choice option under federal guidelines.

 Associated Press
Associated Press